 Burger (Eds.), ‘Oscillations and Travelling Waves in Chemical Systems’, Wiley, New York (1985). Faulkner, ‘Electrochemical Methods’, Wiley, New York (1980) Chapter 6.ĭ. Kubaschewski, ‘Thermochemical Properties of Inorganic Substances - Supplement’, Springer Verlag, Berlin (1977). Knacke, ‘Thermochemical Properties of Inorganic Substances’, Springer Verlag, Berlin (1973). The requirements for successful electroplating. This means that these metals can be used to electroplate other substances. with a positive electrode potential, can be deposited at the cathode during electrolysis. 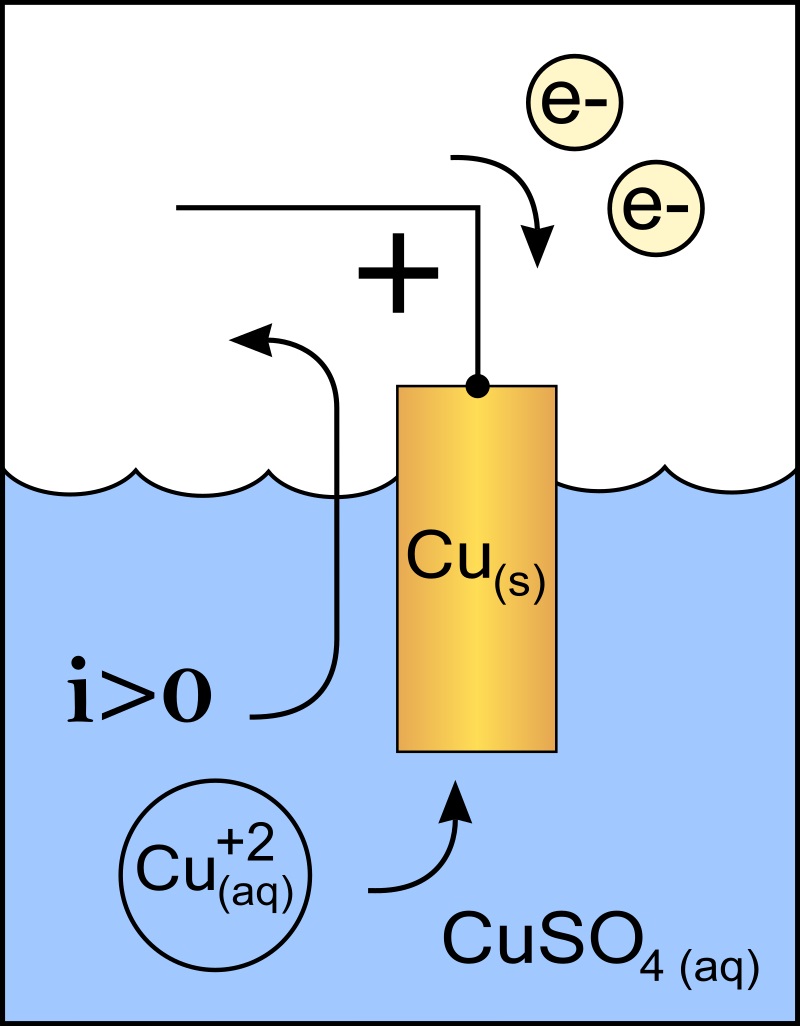
89-2, Hollywood, FL, 15–20 October (1989) Abstract 96, p. Any metal lower than hydrogen in the electrochemical series, i.e. Vissers, ‘The Electrochemical Society Extended Abstracts’, Vol. Sudworth, Chemistry and Industry (1988) 77. Lumsdon, ‘The Electrochemical Society Extended Abstracts’, Vol. At high temperatures (300–400☌) dissolved reagents and products can contribute increasingly to the cathode reaction mechanism. The participation of the solid compound Na 6FeCl 8, which was previously reported to be an intermediate in the charge and discharge processes, has been confirmed, but it does not appear to have any significant adverse effect on the electrochemical performance. Results obtained in a cell containing no liquid electrolyte provide additional support for a solid state mechanism. The following six reactions represent common cathodic reactions along with the conditions in which they generally occur: 1. A predominantly solid state mechanism is thus indicated for the charge and discharge of FeCl 2 cathodes used in high energy sodium cells which operate near 250☌. The reduction half-cell reaction at the cathode depends mainly on environmental conditions. The results suggest that the electrochemical oxidation of iron occurs most readily in the presence of solid NaCl on the electrode, and that solid FeCl 2 is reduced more readily than dissolved Fe(ii) in this electrolyte. 
The polarity of this galvanic cell is such that the anode (supplying the electrons) is. They move from anode to the cathode in the external circuit. Reaction conditions: C anode, Pt cathode, constant current 12 mA, 1a (0.5 mmol), 2a (1.0 mmol). The electrons are supplied by the species getting oxidized. Download scientific diagram Sampling experiment. The reaction at the anode is oxidation and that at the cathode is reduction. Here, the anode is positive and cathode is the negative electrode. The electrochemical behaviour of iron electrodes in molten AlCl 3 : NaCl saturated with NaCl has been studied in the temperature range 175–400 ☌. V <, where is the equilibrium potential of the reaction. The reaction at the anode is oxidation and that at the cathode is reduction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
